Physical Science Reading and Study Worknook Chapter 8
A paper's "Methods" (or "Materials and Methods") department provides information on the study'due south design and participants. Ideally, it should be so articulate and detailed that other researchers can echo the study without needing to contact the authors. You will need to examine this section to determine the written report'due south strengths and limitations, which both touch how the report's results should be interpreted.
Demographics
The "Methods" department normally starts by providing data on the participants, such as age, sexual practice, lifestyle, health status, and method of recruitment. This information will help you decide how relevant the study is to you, your loved ones, or your clients.
Figure 3: Example study protocol to compare two diets
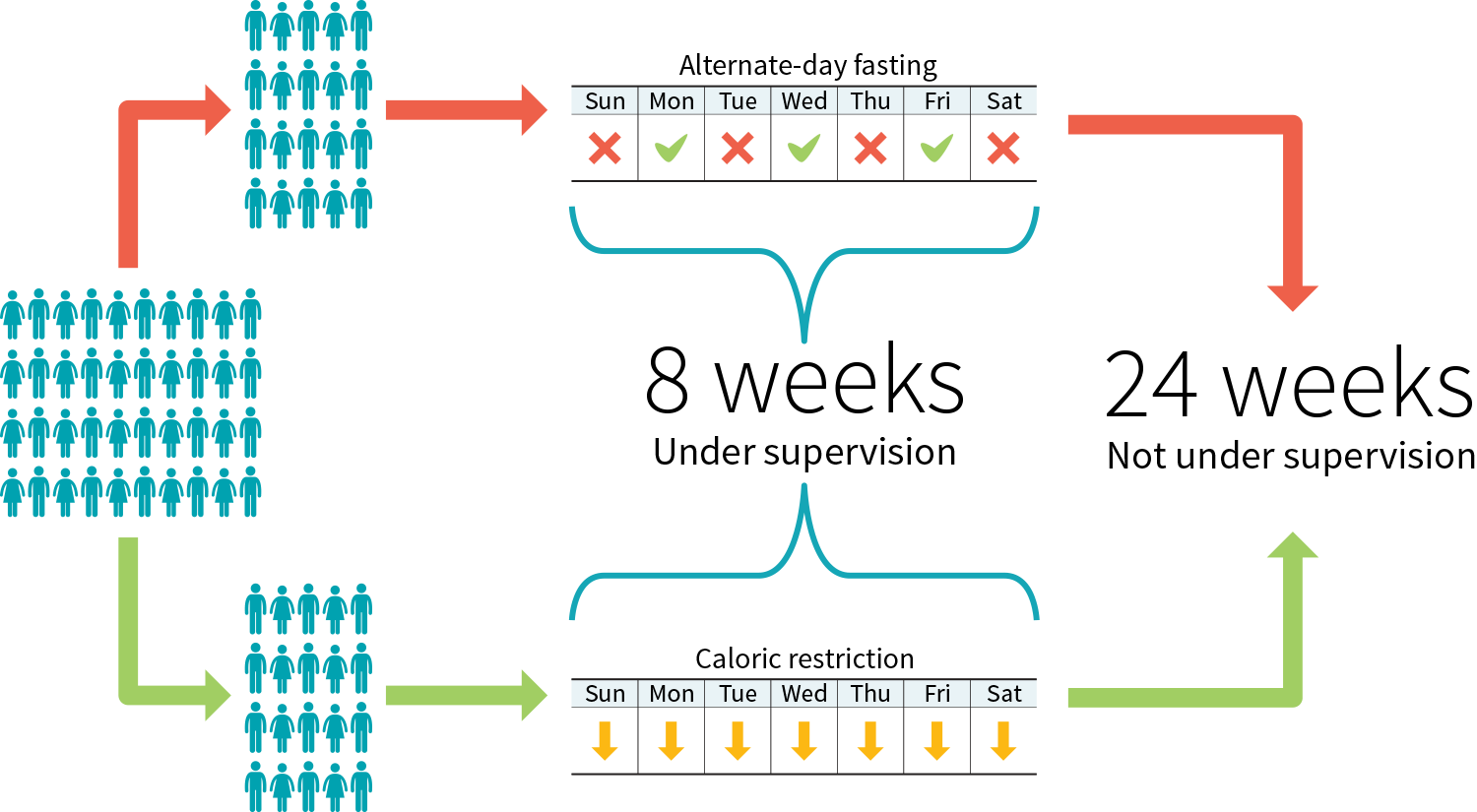
The demographic information tin be lengthy, you might be tempted to skip it, withal it affects both the reliability of the report and its applicability.
Reliability. The larger the sample size of a study (i.due east., the more than participants information technology has), the more reliable its results. Note that a report often starts with more participants than it ends with; diet studies, notably, normally meet a fair number of dropouts.
Applicability. In health and fettle, applicability means that a compound or intervention (i.e., practise, nutrition, supplement) that is useful for ane person may exist a waste of money — or worse, a danger — for another. For case, while creatine is widely recognized equally rubber and constructive, at that place are "nonresponders" for whom this supplement fails to improve exercise performance.
Your mileage may vary, as the creatine instance shows, yet a report'southward demographic information tin help yous assess this study's applicability. If a trial only recruited men, for instance, women reading the study should continue in heed that its results may exist less applicable to them. Also, an intervention tested in college students may yield different results when performed on people from a retirement facility.
Figure four: Some trials are sex-specific

Furthermore, dissimilar recruiting methods will concenter different demographics, and so tin can influence the applicability of a trial. In most scenarios, trialists will utilise some form of "convenience sampling". For instance, studies run by universities will often recruit among their students. However, some trialists volition use "random sampling" to make their trial's results more applicative to the full general population. Such trials are generally called "augmented randomized controlled trials".
Confounders
Finally, the demographic information will usually mention if people were excluded from the study, and if so, for what reason. Virtually often, the reason is the being of a confounder — a variable that would confound (i.e., influence) the results.
For example, if y'all written report the effect of a resistance grooming program on muscle mass, you don't desire some of the participants to take musculus-edifice supplements while others don't. Either you'll want all of them to have the aforementioned supplements or, more likely, yous'll want none of them to take whatsoever.
Likewise, if you study the upshot of a muscle-edifice supplement on muscle mass, you don't want some of the participants to exercise while others exercise non. You lot'll either want all of them to follow the same conditioning programme or, less probable, you'll want none of them to practice.
Information technology is of course possible for studies to take more 2 groups. Y'all could have, for instance, a study on the event of a resistance training plan with the following four groups:
-
Resistance training plan + no supplement
-
Resistance training program + creatine
-
No resistance training + no supplement
-
No resistance training + creatine
But if your study has four groups instead of ii, for each group to keep the aforementioned sample size you need twice as many participants — which makes your study more hard and expensive to run.
When you come correct downwards to it, any differences between the participants are variable and thus potential confounders. That's why trials in mice utilise specimens that are genetically very shut to one another. That's also why trials in humans seldom endeavor to test an intervention on a various sample of people. A trial restricted to older women, for instance, has in effect eliminated age and sex every bit confounders.
As we saw above, with a smashing enough sample size, we can have more groups. We tin even create more groups after the study has run its course, by performing a subgroup analysis. For case, if y'all run an observational written report on the effect of cherry-red meat on thousands of people, you tin later separate the data for "male" from the data for "female" and run a separate analysis on each subset of data. However, subgroup analyses of these sorts are considered exploratory rather than confirmatory and could potentially lead to false positives. (When, for instance, a blood test erroneously detects a disease, it is chosen a false positive.)
Design and endpoints
The "Methods" department will too depict how the study was run. Pattern variants include unmarried-blind trials, in which only the participants don't know if they're receiving a placebo; observational studies, in which researchers only detect a demographic and take measurements; and many more than. (Meet figure two above for more examples.)
More specifically, this is where you will larn about the length of the study, the dosages used, the workout regimen, the testing methods, and so on. Ideally, equally we said, this data should be so clear and detailed that other researchers can repeat the study without needing to contact the authors.
Finally, the "Methods" department tin also brand clear the endpoints the researchers will be looking at. For instance, a written report on the effects of a resistance training program could utilise muscle mass as its primary endpoint (its main benchmark to judge the outcome of the written report) and fat mass, strength performance, and testosterone levels every bit secondary endpoints.
One trick of studies that want to detect an effect (sometimes so that they can serve equally marketing material for a production, but often simply because studies that show an outcome are more than probable to go published) is to collect many endpoints, and then to make the newspaper well-nigh the endpoints that showed an upshot, either by downplaying the other endpoints or by non mentioning them at all. To foreclose such "data dredging/angling" (a method whose devious efficacy was demonstrated through the hilarious chocolate hoax), many scientists push for the preregistration of studies.
Sniffing out the tricks used by the less scrupulous authors is, alas, part of the skills you'll need to develop to appraise published studies.
Interpreting the statistics
The "Methods" section usually concludes with a hearty statistics word. Determining whether an advisable statistical analysis was used for a given trial is an entire discipline, so nosotros advise you don't sweat the details; endeavour to focus on the big motion-picture show.
Commencement, let's clear up two common misunderstandings. You may have read that an effect was significant, only to later observe that it was very small. Similarly, you may have read that no result was found, yet when you read the paper you found that the intervention group had lost more weight than the placebo grouping. What gives?
The trouble is simple: those quirky scientists don't speak like normal people do.
For scientists, significant doesn't hateful of import — information technology means statistically pregnant. An effect is meaning if the information nerveless over the class of the trial would be unlikely if there really was no upshot.
Therefore, an effect can be significant yet very small-scale — 0.2 kg (0.five lb) of weight loss over a year, for instance. More to the point, an result tin be significant however not clinically relevant (meaning that it has no discernible upshot on your wellness).
Relatedly, for scientists, no effect normally means no statistically significant effect. That'due south why y'all may review the measurements collected over the form of a trial and discover an increment or a subtract however read in the conclusion that no changes (or no furnishings) were plant. In that location were changes, merely they weren't significant. In other words, there were changes, but and then modest that they may be due to random fluctuations (they may as well be due to an bodily upshot; we can't know for certain).
We saw earlier, in the "Demographics" section, that the larger the sample size of a study, the more reliable its results. Relatedly, the larger the sample size of a study, the greater its ability to find if small effects are significant. A small change is less likely to be due to random fluctuations when found in a study with a thousand people, let's say, than in a study with ten people.
This explains why a meta-analysis may notice pregnant changes past pooling the data of several studies which, independently, constitute no significant changes.
P-values 101
Most often, an effect is said to be pregnant if the statistical analysis (run past the researchers post-study) delivers a p-value that isn't higher than a certain threshold (set by the researchers pre-study). Nosotros'll call this threshold the threshold of significance.
Agreement how to interpret p-values correctly can be tricky, even for specialists, just hither'due south an intuitive way to think nigh them:
Think about a coin toss. Flip a coin 100 times and you will go roughly a fifty/50 split of heads and tails. Not terribly surprising. But what if you flip this money 100 times and get heads every fourth dimension? Now that's surprising! For the record, the probability of it really happening is 0.00000000000000000000000000008%.
You tin call up of p-values in terms of getting all heads when flipping a coin.
-
A p-value of 5% (p = 0.05) is no more surprising than getting all heads on iv coin tosses.
-
A p-value of 0.5% (p = 0.005) is no more than surprising than getting all heads on 8 coin tosses.
-
A p-value of 0.05% (p = 0.0005) is no more surprising than getting all heads on eleven money tosses.
Reverse to popular belief, the "p" in "p-value" does not stand for "probability". The probability of getting four heads in a row is 6.25%, not v%. If yous desire to catechumen a p-value into coin tosses (technically chosen S-values) and a probability percentage, check out the converter here.
Every bit we saw, an effect is significant if the data collected over the form of the trial would exist unlikely if there actually was no result. Now nosotros tin can add that, the lower the p-value (under the threshold of significance), the more confident nosotros can be that an consequence is significant.
P-values 201
All correct. Off-white warning: nosotros're going to go nerdy. Well, nerdier. Experience free to skip this section and resume reading here.
All the same with us? All right, then — let's get at it. Equally we've seen, researchers run statistical analyses on the results of their study (commonly one assay per endpoint) in guild to decide whether or not the intervention had an effect. They ordinarily make this conclusion based on the p-value of the results, which tells you how probable a upshot at to the lowest degree equally extreme as the one observed would exist if the null hypothesis, among other assumptions, were true.
Ah, jargon! Don't panic, we'll explain and illustrate those concepts.
In every experiment in that location are mostly two opposing statements: the null hypothesis and the culling hypothesis. Let's imagine a fictional study testing the weight-loss supplement "Better Weight" against a placebo. The two opposing statements would await like this:
-
Cypher hypothesis: compared to placebo, Meliorate Weight does non increase or decrease weight. (The hypothesis is that the supplement's effect on weight is null.)
-
Alternative hypothesis: compared to placebo, Better Weight does decrease or increase weight. (The hypothesis is that the supplement has an event, positive or negative, on weight.)
The purpose is to see whether the outcome (here, on weight) of the intervention (here, a supplement chosen "Better Weight") is improve, worse, or the aforementioned as the issue of the control (hither, a placebo, but sometimes the command is another, well-studied intervention; for instance, a new drug tin be studied confronting a reference drug).
For that purpose, the researchers commonly set a threshold of significance (α) before the trial. If, at the end of the trial, the p-value (p) from the results is less than or equal to this threshold (p ≤ α), there is a significant deviation between the effects of the 2 treatments studied. (Recollect that, in this context, significant means statistically significant.)
Figure five: Threshold for statistical significance

The well-nigh ordinarily used threshold of significance is v% (α = 0.05). It ways that if the null hypothesis (i.e., the idea that there was no departure between treatments) is true, and so, after repeating the experiment an infinite number of times, the researchers would get a false positive (i.eastward., would detect a significant effect where in that location is none) at most 5% of the fourth dimension (p ≤ 0.05).
Generally, the p-value is a measure out of consistency between the results of the written report and the idea that the ii treatments have the same effect. Let's see how this would play out in our Better Weight weight-loss trial, where i of the treatments is a supplement and the other a placebo:
-
Scenario 1: The p-value is 0.80 (p = 0.lxxx). The results are more consequent with the null hypothesis (i.e., the idea that at that place is no deviation between the two treatments). We conclude that Better Weight had no significant effect on weight loss compared to placebo.
-
Scenario 2: The p-value is 0.01 (p = 0.01). The results are more than consistent with the alternative hypothesis (i.eastward., the idea that there is a departure betwixt the two treatments). We conclude that Better Weight had a significant effect on weight loss compared to placebo.
While p = 0.01 is a significant result, so is p = 0.000001. So what data do smaller p-values offering united states? All other things being equal, they give us greater confidence in the findings. In our example, a p-value of 0.000001 would give the states greater confidence that Amend Weight had a significant effect on weight change. Simply sometimes things aren't equal betwixt the experiments, making direct comparison between 2 experiment'southward p-values catchy and sometimes downright invalid.
Fifty-fifty if a p-value is significant, remember that a significant effect may not be clinically relevant. Let's say that we institute a pregnant upshot of p = 0.01 showing that Ameliorate Weight improves weight loss. The catch: Improve Weight produced only 0.two kg (0.5 lb) more weight loss compared to placebo after one yr — a deviation besides minor to have any meaningful outcome on health. In this case, though the result is pregnant, statistically, the real-world consequence is too small to justify taking this supplement. (This type of scenario is more likely to take place when the study is large since, as we saw, the larger the sample size of a report, the greater its ability to find if small effects are pregnant.)
Finally, we should mention that, though the most commonly used threshold of significance is 5% (p ≤ 0.05), some studies require greater certainty. For example, for genetic epidemiologists to declare that a genetic clan is statistically meaning (say, to declare that a gene is associated with weight gain), the threshold of significance is unremarkably fix at 0.0000005% (p ≤ 0.000000005), which corresponds to getting all heads on 28 coin tosses. The probability of this happening is 0.00000003%.
P-values: Don't worship them!
Finally, proceed in heed that, while important, p-values aren't the concluding say on whether a written report'due south conclusions are accurate.
We saw that researchers likewise eager to discover an effect in their study may resort to "information angling". They may besides try to lower p-values in various means: for instance, they may run different analyses on the same data and only written report the pregnant p-values, or they may recruit more and more participants until they get a statistically significant result. These bad scientific practices are known as "p-hacking" or "selective reporting". (You can read virtually a real-life example of this hither.)
While a study's statistical analysis usually accounts for the variables the researchers were trying to control for, p-values can likewise be influenced (on purpose or non) by study design, subconscious confounders, the types of statistical tests used, and much, much more. When evaluating the strength of a study'southward blueprint, imagine yourself in the researcher'south shoes and consider how you could torture a written report to make it say what y'all want and advance your career in the procedure.
Source: https://examine.com/guides/how-to-read-a-study/
0 Response to "Physical Science Reading and Study Worknook Chapter 8"
Post a Comment